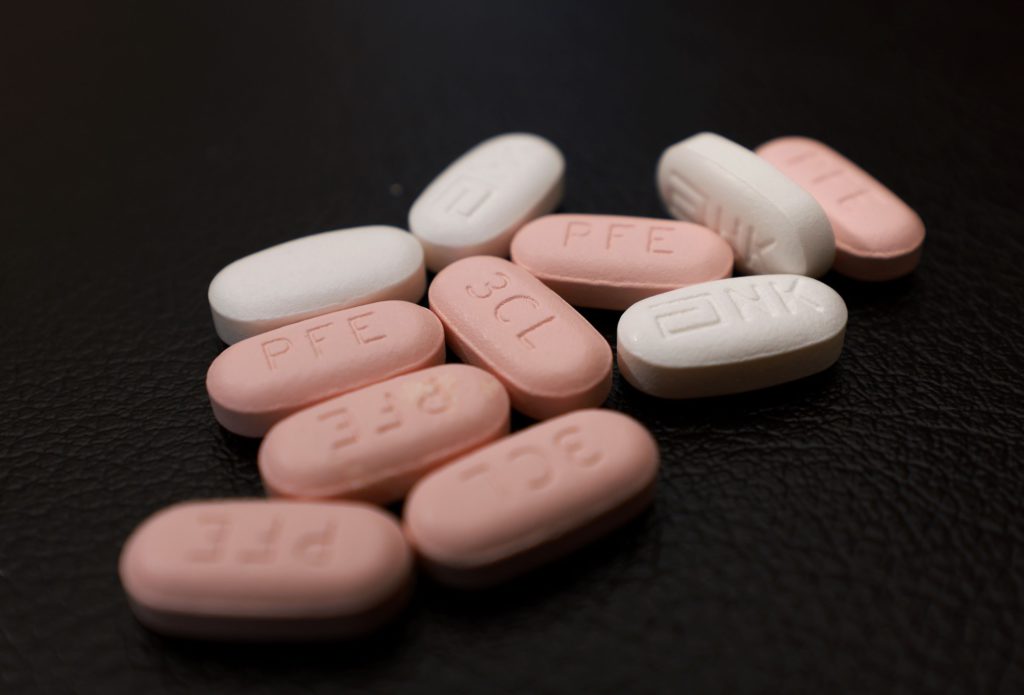
(Bloomberg) — As Covid-19 infections soar across China, a shortage of antiviral medicines like Pfizer Inc.’s Paxlovid appears to be spurring people to turn to the black market.
The country’s abrupt U-turn on Covid Zero earlier this month surprised health experts and residents, as officials appear to have done little planning for the inevitable rise in cases that comes with reopening. That includes easy access to antiviral therapies that can be used by people who test positive and are at higher risk of hospitalization, like the elderly.
The worsening outbreak is boosting demand for such treatments, but Chinese have found the drugs in short supply across the increasingly strained health-care system. People are seeking out online sales channels to source generic versions of the drugs made elsewhere and not approved for sale in China, social media posts and newspaper reports show.
Read more: End of Covid Zero Risks Overwhelming China With Infections
One user on the popular Twitter-like Weibo platform said on Sunday that she was buying generic Paxlovid made in Bangladesh because she had an elderly relative and couldn’t get a hold of any in China. She said in the post, which has been removed, that while she’d heard China had imported tens of thousands of boxes of Paxlovid, ordinary residents weren’t able to access the potentially life-saving medicine.
The same day, another Weibo user called for easier and quicker access to the pills. The poster shared a picture of an advertisement for generic versions of Paxlovid made in India that can be shipped the same day as payment and arrive in two-to-three weeks.
Those that can get their hands on antivirals are paying huge premiums. The 21st Century Business Herald reported an unidentified white-collar worker in Guangdong paid 5,800 yuan ($830) to a Hong Kong agent for a box of Paxlovid, more than double the official price in mainland China. The Paper, a Shanghai-based publication, reported purchasing agents have sold more than 50,000 boxes of foreign generic antivirals since the government’s first tentative steps toward easing Covid Zero in November.
Hu Xijin, the retired editor-in-chief of state-backed tabloid the Global Times and a vocal pro-government commentator, joined the chorus lamenting the high price and noted dissatisfaction among Chinese.
It’s unclear whether China is prepared to satisfy the huge demand, according to Siddharth Sridhar, clinical assistant professor in the University of Hong Kong’s department of microbiology. “Even if there is enough Paxlovid for China’s large population, a distribution infrastructure to get these pills to patients in time isn’t something that can be set up overnight,” he said.
How Will China’s Covid Pivot Play Out? Your Questions Answered
When China was still trying to keep out the virus, securing adequate supplies of antivirals was described by top health officials like Liang Wannian as essential before the country would consider reopening. The sudden about-turn on Covid means that the under-vaccinated elderly population that benefit most from such drugs are highly vulnerable.
The supply strain may also be intensified by heightened concern among Chinese about the dangers of Covid after three years of propaganda that painted the virus as exceptionally deadly. With the government’s official stance changing — one top medical adviser likened it to a cold — so has public messaging, and state media have published articles warning that not everyone needs to take antivirals. Earlier this week, a top infectious disease doctor said patients needing the therapies should take them under medical supervision and they’re not suitable for people already taking certain medications.
Regulatory Maze
Increasing supply is also complicated, and there’s little transparency over how much is available in China.
Paxlovid is the only foreign Covid medicine approved by China’s regulator for nationwide use, though AstraZeneca’s preventative antibody drug Evusheld can be accessed in a medical pilot zone in the southern island of Hainan. State-owned China Meheco Co. said last week it reached a deal with Pfizer to import and distribute the latter’s antiviral. Pfizer in August also announced a deal with Zhejiang Huahai Pharmaceutical Co., to make ingredients of Paxlovid in the country.
Pfizer said in a statement that it’s collaborating with all stakeholders to secure an adequate supply of Paxlovid in China and remains committed to fulfilling the treatment needs of patients in the country and around the world. It said it’s committed to bringing medicines and therapies including Paxlovid that will benefit Chinese patients as soon as possible.
As a prescription medication, Paxlovid’s purchase channel will strictly align with government regulations, Pfizer said. The company continues to evaluate options to increase capacity, including opportunities to expand manufacturing capabilities, increase supplier base for key materials and contract manufacturing options for its supply chain, it said.
Meheco didn’t respond to an email or phone call seeking comment. Media outlet Yicai reported Tuesday, citing a phone call in which a journalist posed as an investor, that Meheco didn’t expect sales of Paxlovid to surge despite the massive waves of infection.
Merck & Co.’s molnupiravir isn’t approved, though in September it said it struck a deal with a state-owned drugmaker to import and market the pill.
Running Out
Even what China can produce domestically doesn’t always make it to its own residents.
As part of efforts to provide generic low-cost antivirals to developing countries, the United Nations-backed Medicines Patent Pool program in March authorized five Chinese companies to make Paxlovid, but exclusively for export. The group includes Huahai, which struck a separate deal with Pfizer to make Paxlovid for China’s domestic market.
A representative in Huahai’s investor relations department said it hasn’t started making generic versions of Paxlovid under the MPP agreement, and declined to comment on whether the company has started production of Paxlovid ingredients for the China market under the deal with Pfizer.
Some local companies are developing antivirals too. Henan Genuine Biotech Co. has received approval for the drug Azvudine, an HIV treatment that doctors can now prescribe for Covid patients. An antibody cocktail therapy developed by Brii Biosciences Ltd. has also been given the green light but it requires infusion at hospitals.
For now, there’s no end in sight to the antivirals shortage. On Friday, a hospital in the northeastern city of Jinan began offering Azvudine, sparking an endless stream of inquiries and buyers, Qilu Evening News reported.
While the hospital limited each person to two boxes, by Sunday it had sold all of its 5,000 pill supply.
–With assistance from Nacha Cattan.
(Updates with statement from Pfizer under ‘Regulatory Maze’ sub-heading)
More stories like this are available on bloomberg.com
©2022 Bloomberg L.P.